Electron Configuration
Bohr Rutherford Diagrams and ions YouTube
0:00 / 5:40 Bohr-Rutherford Diagram of NaCl (sodium chloride, table salt) chemistNATE 260K subscribers Subscribe Subscribed Share 6.7K views 3 years ago NaCl, sodium chloride, is an IONIC.

Potassium Bohr Model Diagram, Steps To Draw Techiescientist
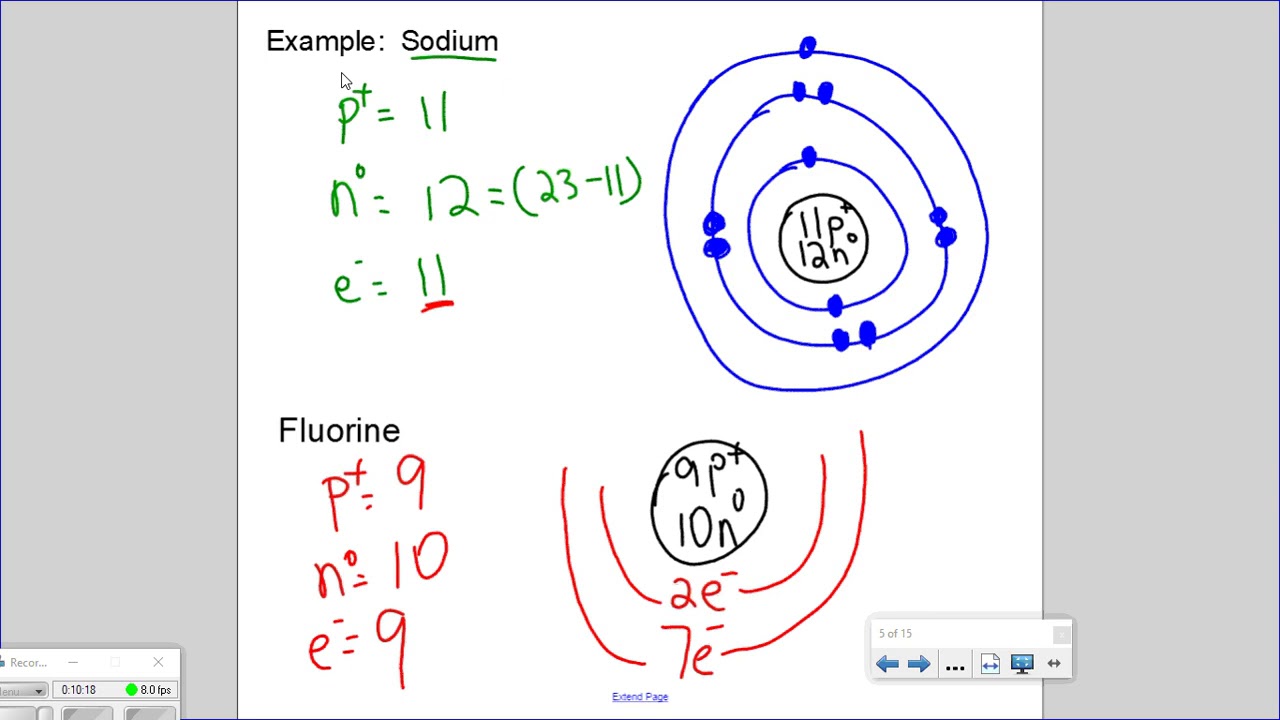
Bohr's model of the atom can be combined with Rutherford's model in diagrams that summarize the numbers and positions of all three subatomic particles. For example, consider the following diagram for Phosphorous: There are certain rules to follow when drawing these diagrams: A circle is drawn in the center to represent the nucleus of the atom.
Give an example of an atom and an ion, and draw a BohrRuthe Quizlet
In atomic physics, the Bohr model or Rutherford-Bohr model of the atom, presented by Niels Bohr and Ernest Rutherford in 1913, consists of a small, dense nucleus surrounded by orbiting electrons.
How To Draw A Bohr Diagram Wiring Diagram
Bohr-Rutherford diagrams are simple atomic models that show the number of electrons in each shell of an atom. While they are a major simplification of what really happening in an atom, they can be useful to help with visualizing electrons orbiting a nucleus. Drawing Bohr-Rutherford diagrams is super easy using the following steps:

How To Draw A Bohr Model For An Ion
The plum pudding model was suggested as the first atomic model by J.J Thomson where he suggested that the atom was a sea of positive charge that surrounded small negative electrons. Ernest Rutherford. Ernest Rutherford was a British physicist who by experimenting with gold foil and alpha particles found that there was a large central mass at.
PPT How to Draw Bohr Diagrams PowerPoint Presentation, free download
Bohr's model calculated the following energies for an electron in the shell, n. . : E ( n) = − 1 n 2 ⋅ 13.6 eV. Bohr explained the hydrogen spectrum in terms of electrons absorbing and emitting photons to change energy levels, where the photon energy is. h ν = Δ E = ( 1 n l o w 2 − 1 n h i g h 2) ⋅ 13.6 eV.

Bohr's Atomic Model — Overview & Importance Expii
Here are a few examples of Bohr Rutherford diagrams for different elements: 1. Hydrogen (H) Hydrogen has one electron. In the Bohr Rutherford diagram, the nucleus is represented as a small dot in the center, and the electron is shown in the first energy level surrounding the nucleus. It is represented by a single dot or circle. 2. Oxygen (O)
How To Draw Bohr Models For Ions
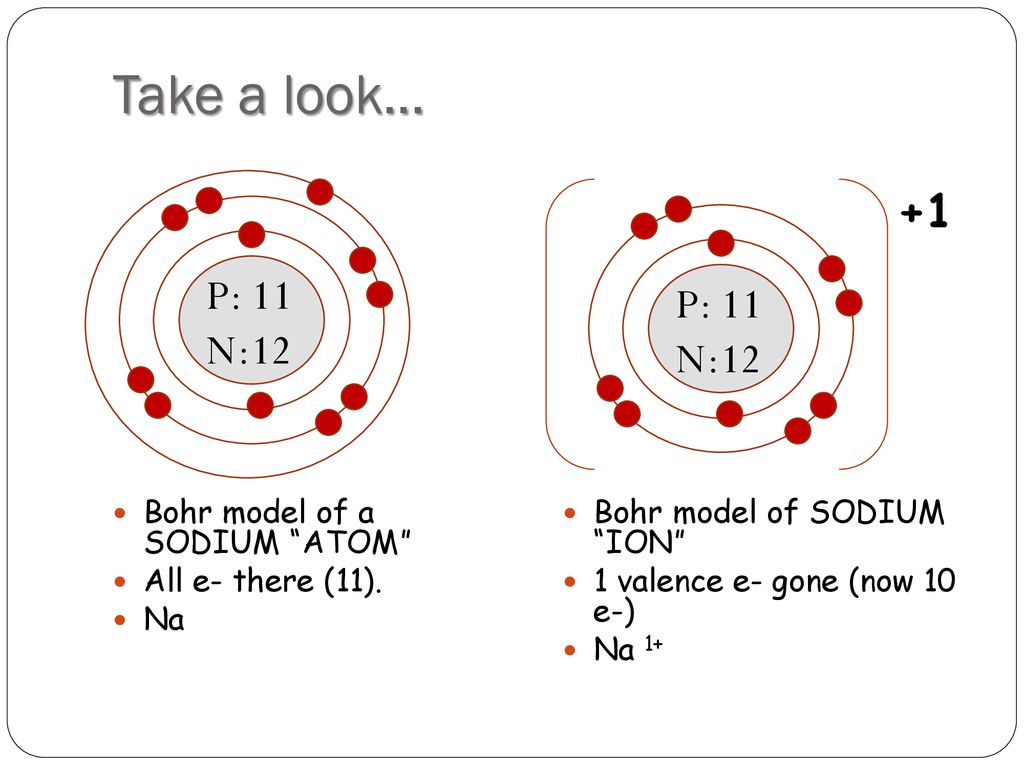
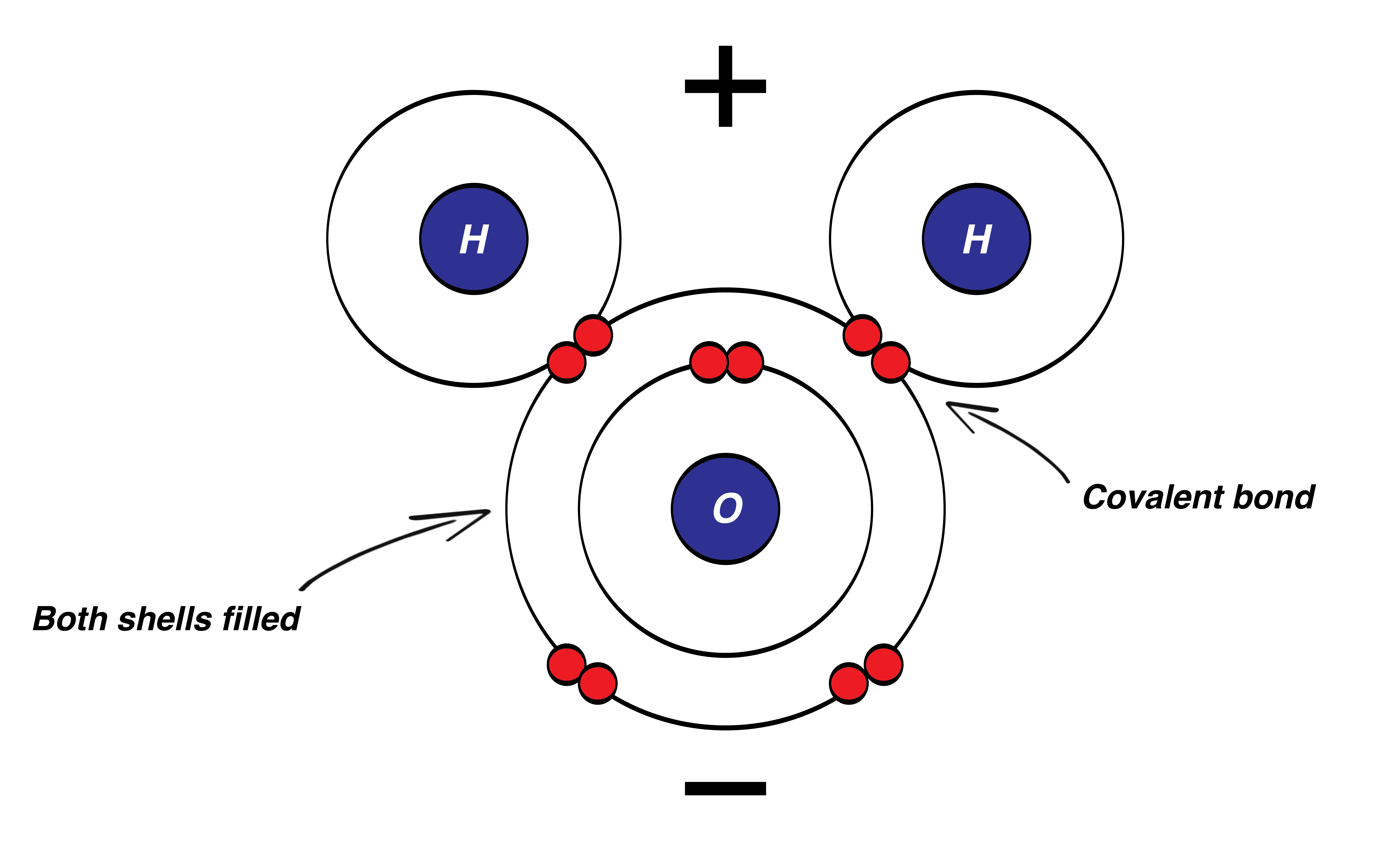
Manish Bhardwaj. 5.6: Bohr Diagrams of Atoms and Ions is shared under a not declared license and was authored, remixed, and/or curated by LibreTexts. Bohr diagrams show electrons orbiting the nucleus of an atom somewhat like planets orbit around the sun. In the Bohr model, electrons are pictured as traveling in circles at different shells,..
Lesson 4 THE STRUCTURE OF THE ATOM WillowWood Lessons
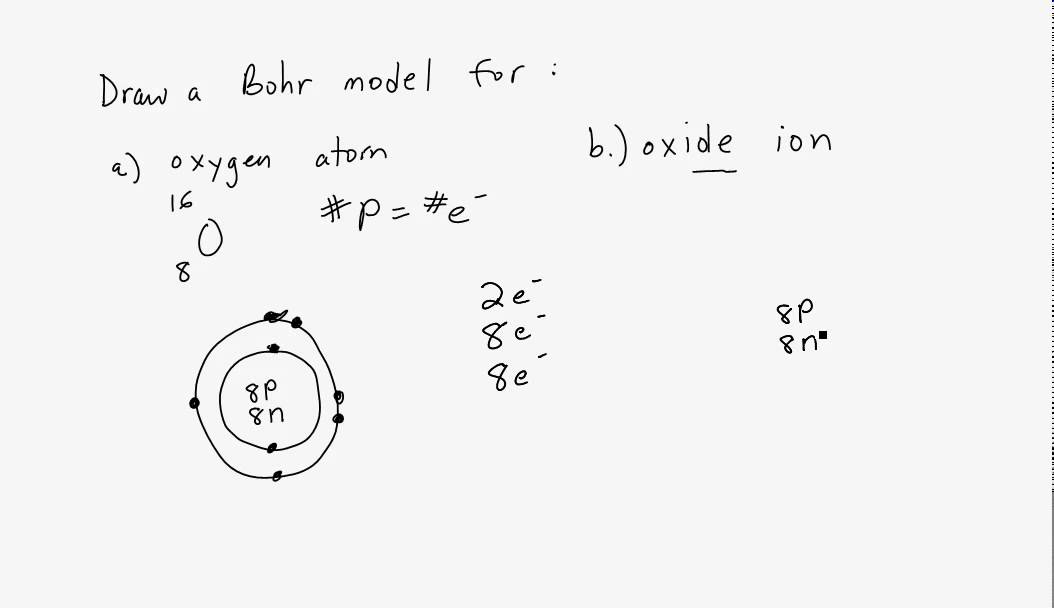
Steps to Draw the Bohr Model of Oxygen. The oxygen atom belongs to the 16 th group of the periodic table. The information that we can derive from the above-mentioned Oxygen box is as follows: • The atomic number of Oxygen is 8. • The electronic configuration of Oxygen is [He] 2s 2 2p 4. • The chemical symbol of Oxygen is O.
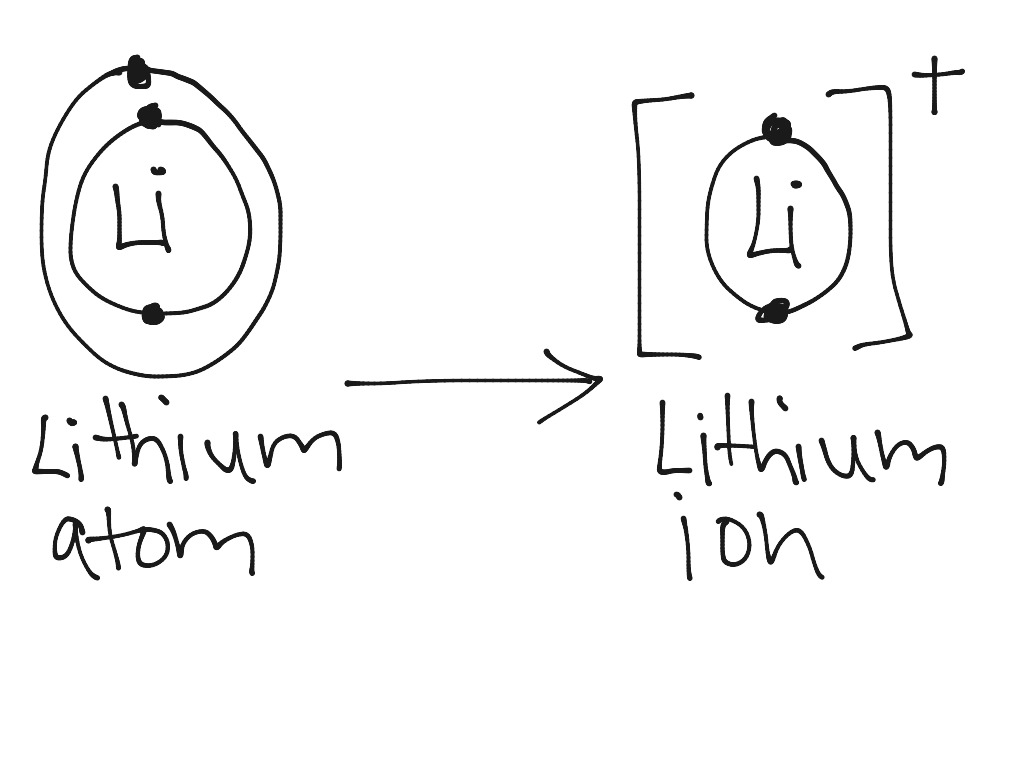
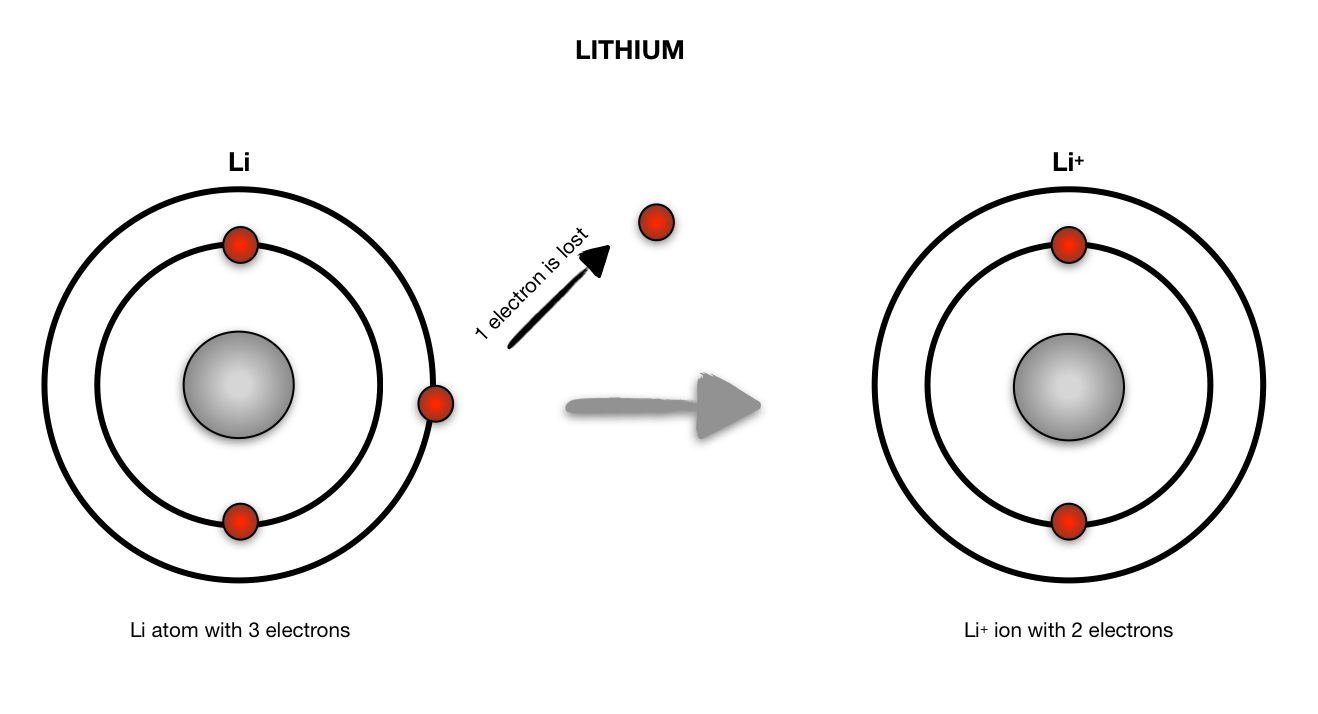
Lithium atom to lithium ion Science, Chemistry, Chemical Bonds ShowMe
Bohr diagrams for Ionshttp://www.BCLearningNetwork.com.0:01the0:02in0:04this video shows you how to drop or diagrams for Alliance0:08chilling protons neutron.
Bohr Rutherford Diagram For Nitrogen
How to draw the Bohr-Rutherford Diagram for Oxygen. 2 electrons can go in the first shell, 8 in the second, 8 in the third, and so on.
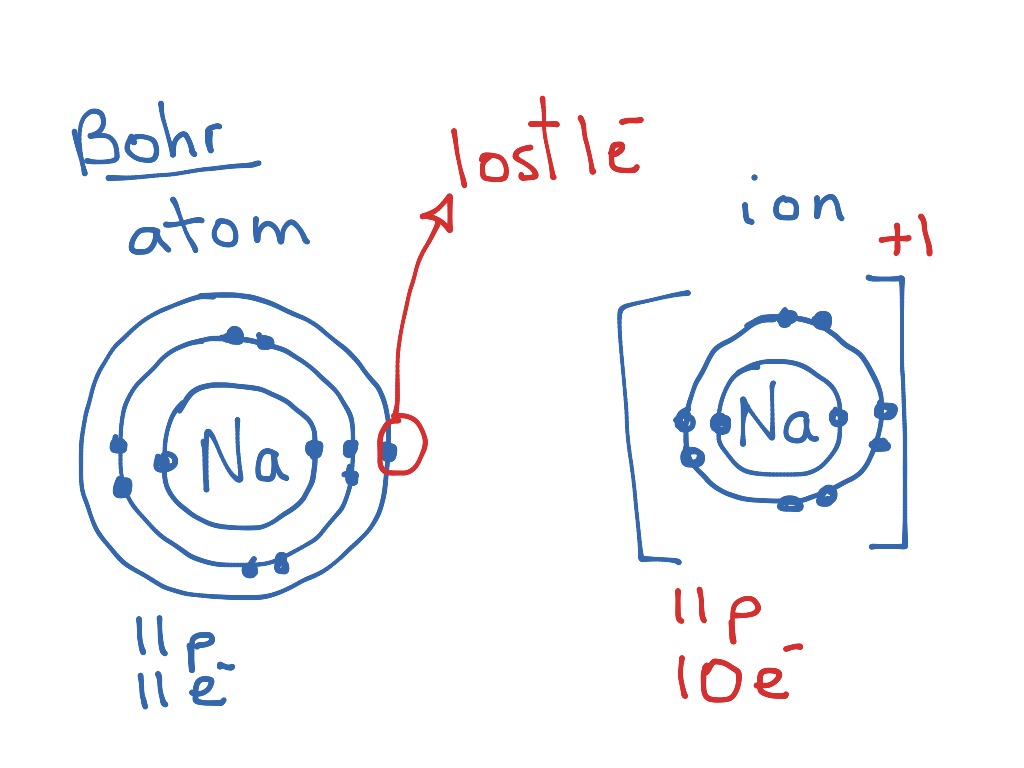
[DIAGRAM] Bohr Diagram For Ion
Since Bohr's model involved only a single electron, it could also be applied to the single electron ions He +, Li 2 +, Be 3 +, and so forth, which differ from hydrogen only in their nuclear charges, and so one-electron atoms and ions are collectively referred to as hydrogen-like or hydrogenic atoms. The energy expression for hydrogen-like.
bohr rutherford diagrams lithium
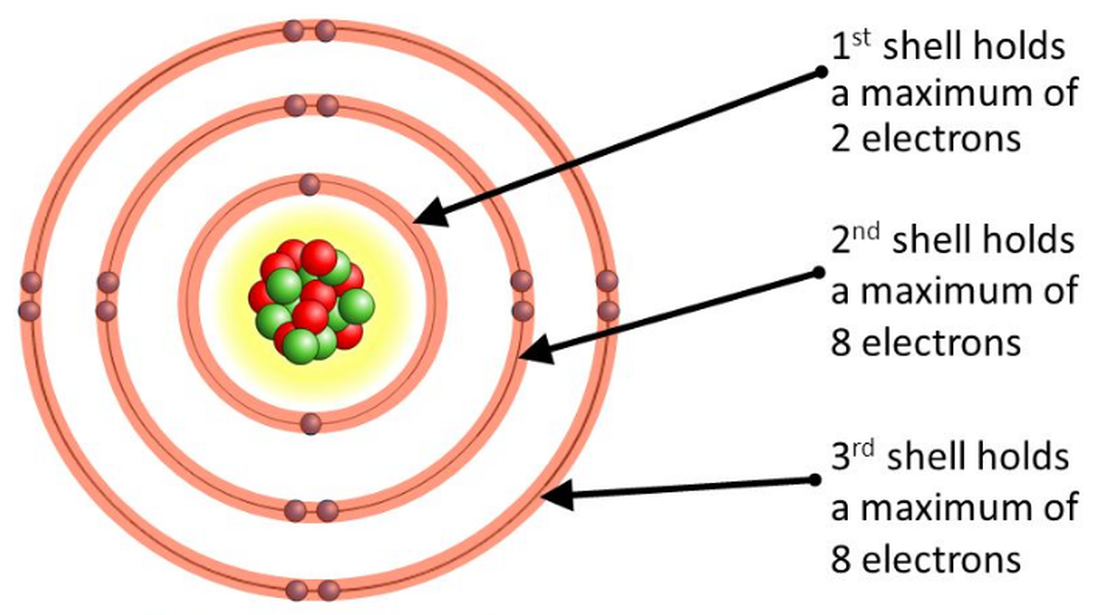
Niels Bohr and Ernest Rutherford studied it and found the number of electrons each orbit needed to maintain the balance between the forces. They drew a diagram to represent the electrons in each orbit. Each orbit in the atom is called an energy shell or energy level. The shell nearest to the nucleus can hold a maximum of 2 electrons.

Image result for silicon atomic model
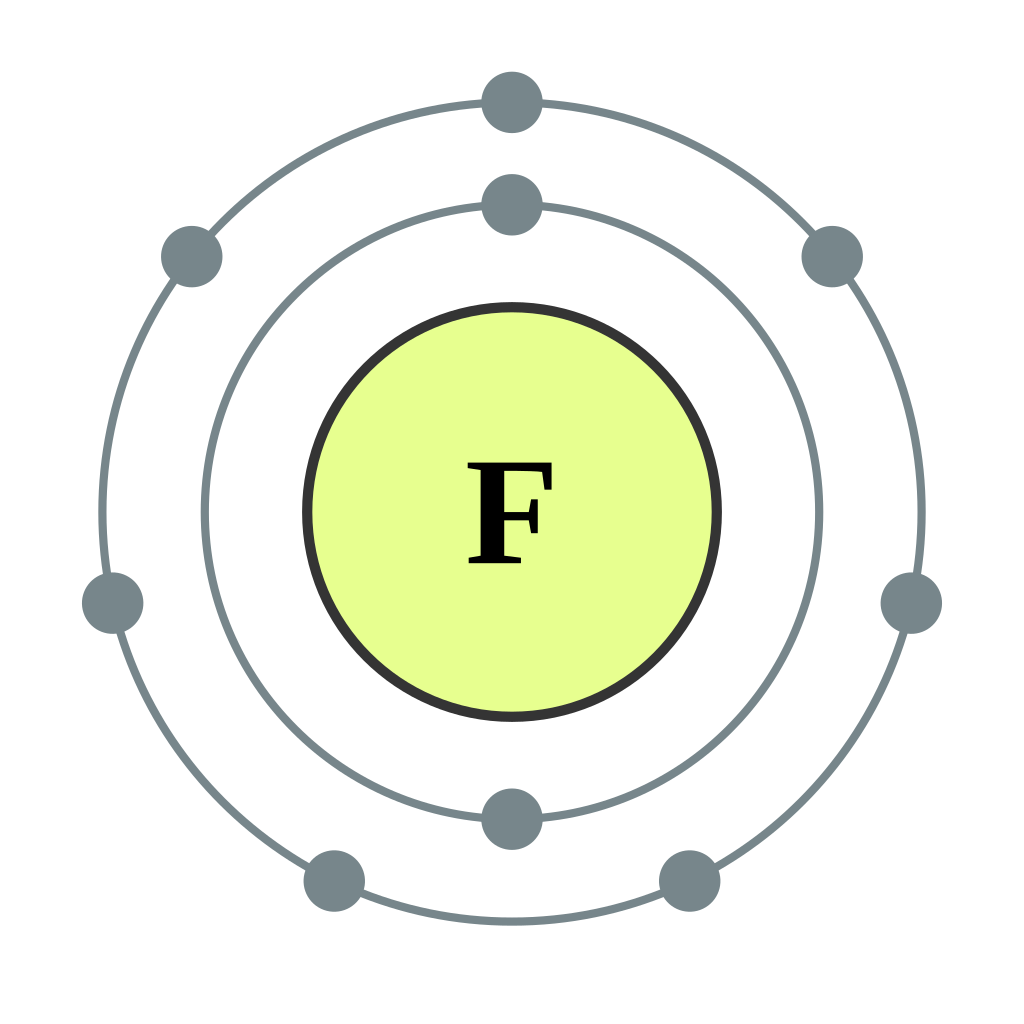
Bohr diagrams show electrons orbiting the nucleus of an atom somewhat like planets orbit around the sun. In the Bohr model, electrons are pictured as traveling in circles at different shells, depending on which element you have. Figure 2 2 contrast the Bohr diagrams for lithium, fluorine and aluminum atoms. The shell closest to the nucleus is.
_0.jpg?itok=RuBFwyf1)
Le modèle atomique de RutherfordBohr Alloprof
Using the Bohr model, determine the energy, in electron volts, of the photon produced when an electron in a hydrogen atom moves from the orbit with n = 5 to the orbit with n = 2. Show your calculations. Using the Bohr model, determine the lowest possible energy, in joules, for the electron in the Li 2+ ion.
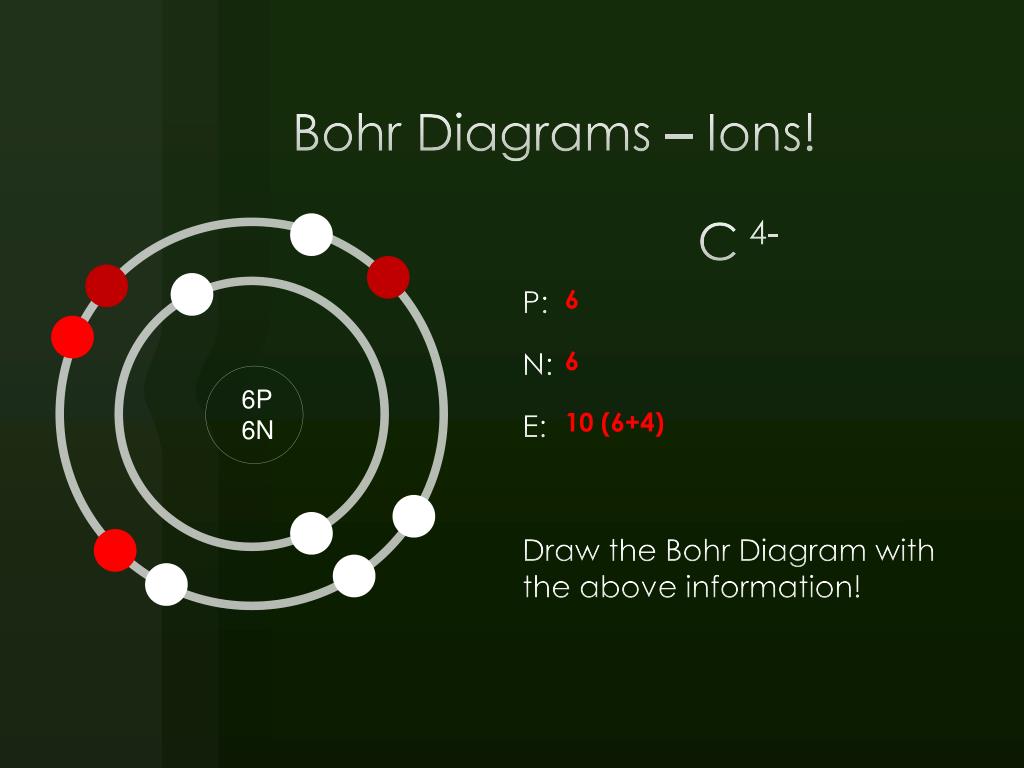
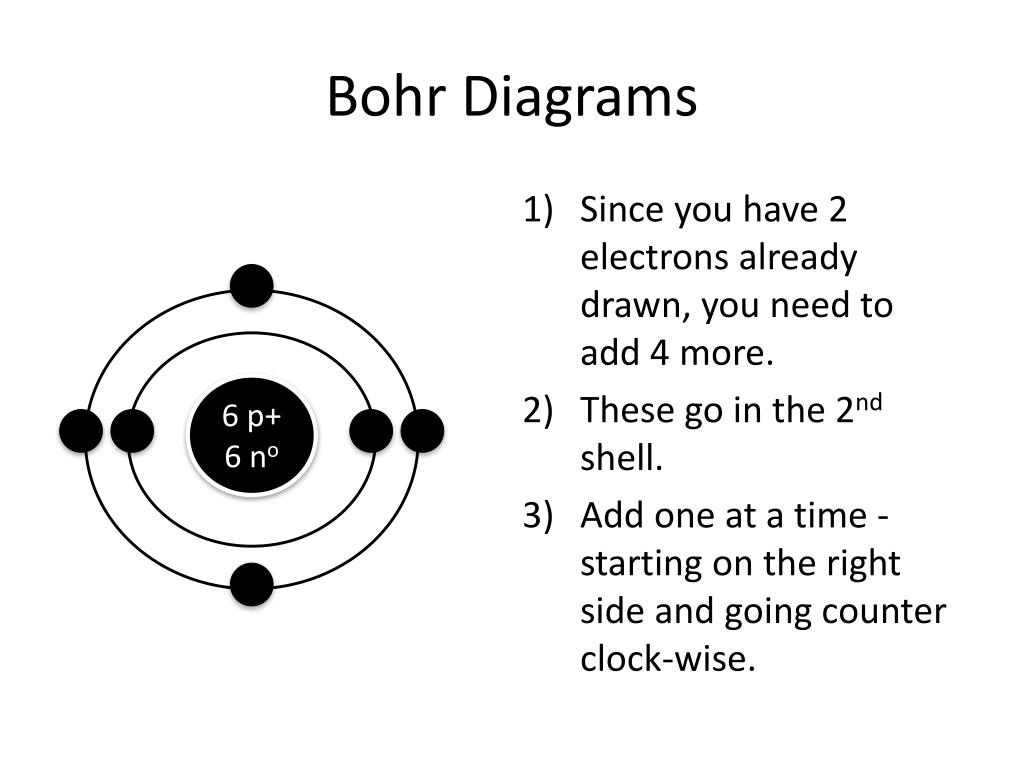
PPT Bohr Diagrams PowerPoint Presentation, free download ID3786522
This chemistry tutorial video walks you through how to draw a Bohr model, Bohr diagram, or planetary model for an atom or ion. The protons and neutrons are p.